
Raltegravir versus efavirenz in antiretroviral-naive pregnant women living with HIV (NICHD P1081): an open-label, randomised, controlled, phase 4 trial - The Lancet HIV

Biostats Masters Student Jinyi Zhou Receives Poster Award | Duke Department of Biostatistics and Bioinformatics
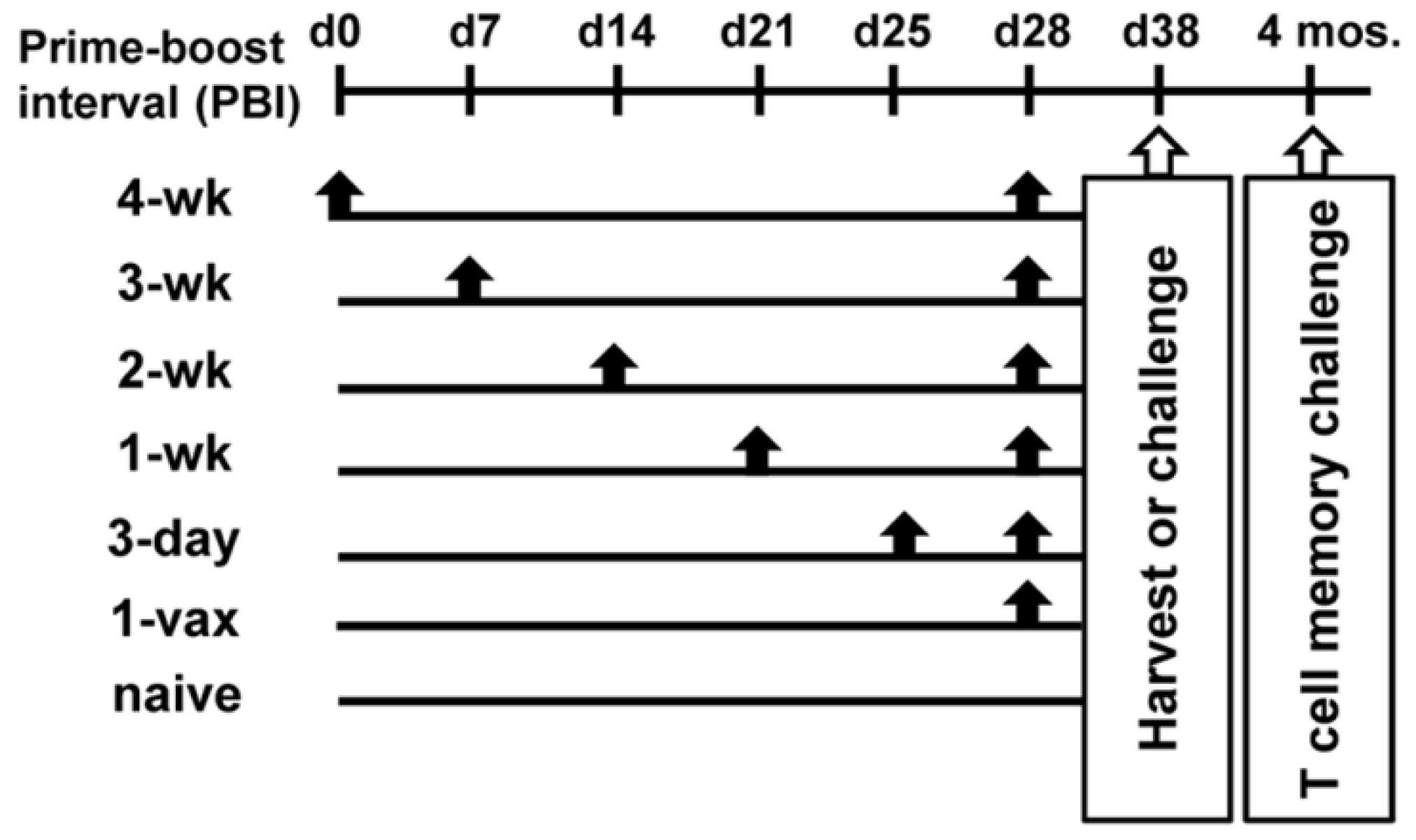
Cancers | Free Full-Text | Investigation of the Optimal Prime Boost Spacing Regimen for a Cancer Therapeutic Vaccine Targeting Human Papillomavirus | HTML

Incoming HIV virion-derived Gag Spacer Peptide 2 (p1) is a target of effective CD8+ T cell antiviral responses - ScienceDirect
Difference in Restricted Mean Survival Time for Cost-Effectiveness Analysis Using Individual Patient Data Meta-Analysis: Evidence from a Case Study | PLOS ONE













